Apr 9, 2026
Industry Best Practices for Change Activation in Pharmaceutical Companies
by
Nellie Wartoft
CEO & Founder
You need fast adoption to keep up with pharmaceuticals’ latest advancements, but also precision in maintaining quality, safety, and global coordination.
Change Activation makes that scalable.
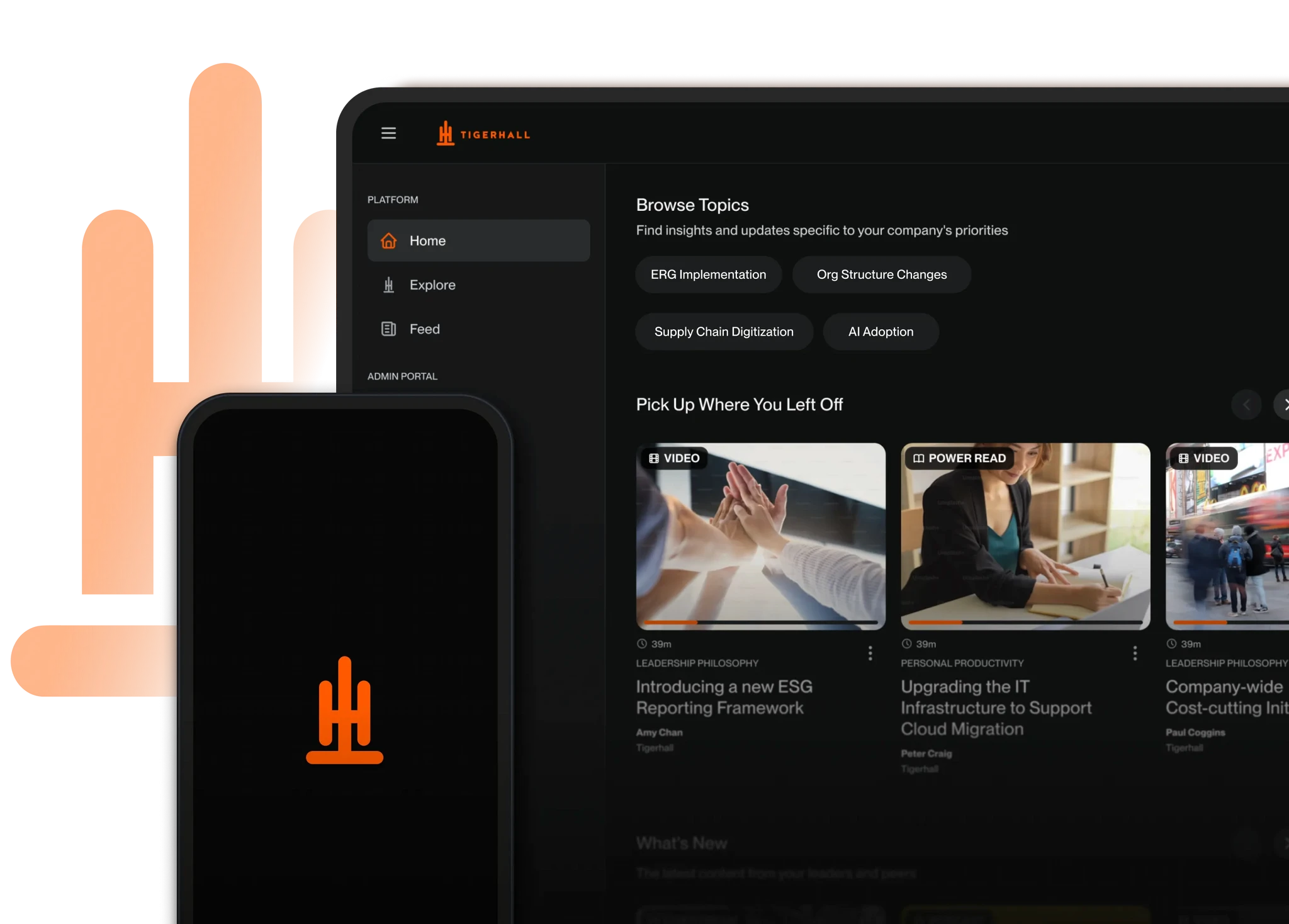
See how the Change Activation Operating Model mobilizes leaders and teams around high-impact transformation initiatives—without compromising control or performance.
You’ll learn how to:
Activate regulatory and product updates quickly and consistently across teams, manufacturing sites, and markets
Flag teams running on outdated SOPs before they impact audits or regulatory submissions
Use the Tigerhall platform to deliver targeted, in-the-workflow guidance that reinforces new protocols and priorities long after launch
Follow along with the deck here.
Tigerhall is the only platform built on the Change Activation Operating Model. Explore now: tigerhall.com/schedule-a-demo